Digital Support for Breast Cancer Survivorship
Understanding Patient Needs and the Potential Role of the RESIL.IO Platform
White paper
RESIL.IO
March 20, 2026
Abstract
Background: Hormonal therapy is a cornerstone of treatment for hormone receptor–positive breast cancer, significantly reducing recurrence risk and improving survival outcomes. However, long-term adherence to endocrine therapy remains a major challenge due to persistent side effects such as fatigue, joint pain, hot flashes and sleep disturbances. These symptoms can significantly affect quality of life and may lead to treatment interruption or discontinuation [1,2]
Digital health interventions represent a promising approach to support patients during long-term cancer treatments by providing symptom monitoring, patient education and lifestyle support. [3].
Objective: To evaluate patient-reported needs, adherence challenges, and feature preferences for the RESIL.IO digital health intervention designed to support women undergoing hormonal cancer treatment.
Methods: A cross-sectional survey of 118 women including 72 currently or previously on hormone therapy, assessed demographics, treatment history, adherence, side effects, and health app usage. Open-ended responses captured unmet needs and desired functionalities. Descriptive statistics and thematic analysis were applied.
Results: Most participants were aged 45–60 (58.5%) and had undergone prior treatment including surgery (71%) and radiotherapy (70%). Fatigue (70%) and joint pain (59%) were the most common side effects. Mean self-reported quality-of-life impact was 6.87/10. Thirteen participants (19%) modified or interrupted treatment without medical consultation [2]. Most respondents (60%) had never used a health app, but 81% expressed willingness to use RESIL.IO regularly. Key desired features included symptom tracking (66%), reliable medical information (37%), and lifestyle support (mental, nutrition, exercise; 43%)
Conclusion: Hormonal therapy in breast cancer patients is associated with significant unmet needs regarding side effects management and adherence support. Digital interventions like RESIL.IO can play a critical role in improving quality of life and adherence. Future studies should evaluate clinical outcomes and integration into standard care pathways.
1. Breast Cancer Survivorship: An Ongoing Challenge
Breast cancer is the most common cancer among women worldwide, with more than 2.3 million new cases diagnosed annually. Advances in screening, diagnosis and treatment have significantly improved survival rates over the past decades. As a result, the number of breast cancer survivors continues to increase.
Approximately 70% of these cancers are hormone receptor–positive and require long-term endocrine therapy.
While hormonal therapy significantly reduces recurrence risk and improves survival, adherence remains a major challenge. Studies show that up to 30–50% of patients discontinue treatment prematurely, increasing the risk of recurrence and mortality [1].
For patients with hormone receptor–positive breast cancer, endocrine therapy plays a critical role in reducing the risk of recurrence and improving long-term survival. Treatments such as tamoxifen or aromatase inhibitors are typically prescribed for five to ten years following primary treatment.
Despite their proven efficacy, these therapies are frequently associated with persistent side effects [1,2] including:
- fatigue
- joint pain and musculoskeletal symptoms
- hot flashes
- sleep disturbances
- mood changes.
These symptoms can substantially impact daily life and represent one of the leading causes of treatment discontinuation.
Several studies have shown that up to 30–50% of patients discontinue endocrine therapy prematurely, which may increase the risk of cancer recurrence and negatively affect long-term outcomes.
Improving adherence to long-term treatments therefore represents a critical challenge in breast cancer survivorship.
2. The Potential of Digital Health Interventions
Digital health technologies are increasingly recognized as valuable tools for supporting patients throughout their treatment journey.
Mobile health applications and digital therapeutic platforms can provide patients with:
- symptom monitoring tools
- educational resources
- lifestyle and behavioral support
- improved communication with healthcare professionals.
Such interventions may contribute to improved patient engagement, better symptom management and enhanced adherence to treatment.
However, the development of effective digital health solutions requires a clear understanding of patient needs, expectations and potential barriers to adoption.
The present survey was conducted to better understand the experiences of women undergoing hormonal therapy and to explore their expectations regarding digital support solutions.
3. Study Objectives
The objectives of this survey were to:
- Identify adherence challenges faced by women undergoing hormonal therapy
- Capture patient-reported side effects and their impact on daily life.
- Understand expectations and preferences for digital health solutions, specifically RESIL.IO.
- Explore prior experience with health apps to inform adoption strategies.
4. Methodology
Study Design
Participants: 72 women currently or previously undergoing hormone therapy; 69 provided age data.
Survey Instrument: K-Search; a structured questionnaire captured demographics, treatment history, hormone therapy adherence, , side effects, quality-of-life impact (1–10 scale), health app usage, and feature preferences. Open-ended responses explored unmet needs and desired functionalities.
Ethics: Participation was voluntary; data were anonymized.
Data Analysis: Descriptive statistics (frequencies, percentages, means) summarized quantitative data. Qualitative responses were analysed thematically to identify recurrent patterns and priorities.
A cross-sectional survey was conducted among women diagnosed with breast cancer, recruited via social media patient communities or RESIL.IO communities.
Participants
A total of 118 women participated in the survey.
The final analytical sample included 72 participants who were either currently undergoing or had previously received hormonal therapy.
Data Collection
The questionnaire collected information regarding:
- demographic characteristics
- treatment history
- side effects experienced during hormonal therapy
- perceived impact on quality of life (1–10 scale),
- treatment adherence behaviors
- prior experience with health applications
- expectations regarding digital health solutions.
Open-ended questions allowed participants to describe their experiences and expectations in their own words.
5. Results
Demographic Characteristics
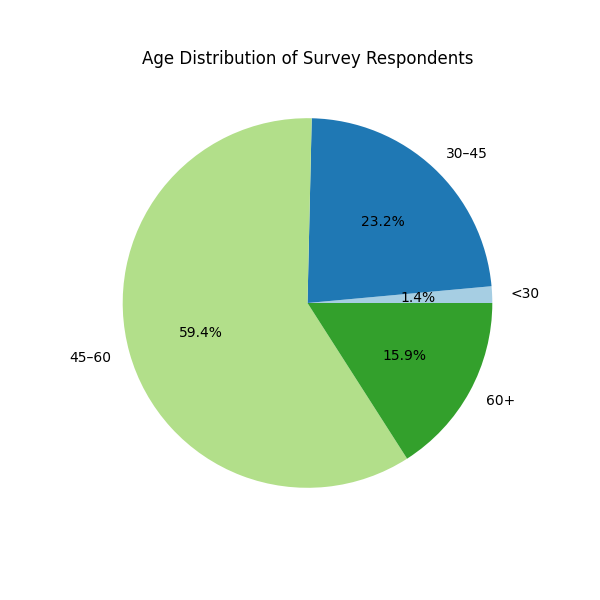
The age distribution of respondents was as follows:
- 45–60 years: 58.5%
- 30–45 years: 23%
- over 60 years: 16%
- under 30 years: 1%.
Most participants had received multiple types of cancer treatment:
- surgery: 71%
- radiotherapy: 70%
- adjuvant chemotherapy: 33%
- neoadjuvant chemotherapy: 27%.
Side Effects Associated with Hormonal Therapy
Participants reported several persistent side effects during hormonal therapy.
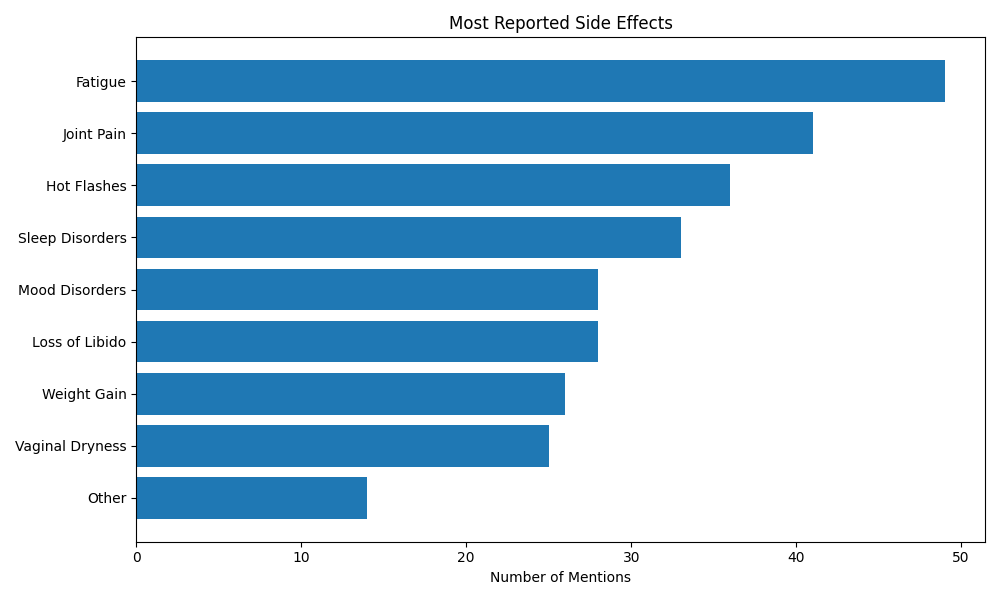
The most frequently reported symptoms were:
- fatigue (70%)
- joint pain (59%)
- hot flashes (52%)
- sleep disturbances (48%).
These symptoms were frequently described as having a substantial impact on daily functioning and overall well-being.
Impact on Quality of Life
Participants were asked to rate the impact of hormonal therapy on their quality of life on a scale from 1 to 10.
The average reported impact score was 6.87, indicating a moderate to high perceived burden associated with treatment.
Treatment Adherence
Among the participants receiving hormonal therapy, 19% reported having modified or interrupted their treatment without consulting a physician.
These findings highlight the challenges associated with long-term treatment adherence.
Side effects, fatigue and psychological distress were among the most frequently cited reasons for treatment modification.
Digital Health Usage and Expectations
The survey also explored participants’ experience with digital health applications.
Results showed that:
- 60% of participants had never used a health application
- despite this, 81% expressed interest in using a digital platform such as RESIL.IO on a regular basis.
Participants identified several features they would find particularly useful:
- symptom tracking (66%)
- communication with healthcare professionals (46%)
- lifestyle guidance including nutrition, mental health and physical activity (43%)
- access to reliable medical information (37%).
These results suggest a strong demand for structured digital support solutions in breast cancer survivorship.
6. The RESIL.IO Digital Support Platform
RESIL.IO is a digital health platform designed to support women undergoing long-term breast cancer treatment.
The platform aims to address several unmet needs identified in the survey.
Side effects Monitoring
RESIL.IO enables patients to track treatment-related side effects over time, helping them better understand their status and identify changes that may require attention.
Such monitoring may also facilitate communication between patients and healthcare providers.
Evidence-Based Lifestyle Support
Lifestyle factors play a key role in overall well-being during and after cancer treatment.
The platform integrates guidance on:
- nutrition
- physical activity
- mental health.
These components aim to help patients manage symptoms and improve their quality of life.
Patient Education
Access to reliable medical information is essential for reducing anxiety and supporting informed decision-making.
RESIL.IO provides educational content approved and reviewed by medical expert designed to help patients better understand their treatment and manage its potential side effects.
Patient Empowerment
By providing tools for monitoring, education and self-management, the platform seeks to empower patients to take an active role in their care journey.
7. Discussion
The results of this survey highlight the significant burden associated with hormonal therapy in breast cancer survivors.
Fatigue, joint pain and sleep disturbances were among the most commonly reported symptoms and were frequently associated with a reduced quality of life.
The finding that nearly one in five participants reported modifying or interrupting treatment without medical consultation underscores the importance of improving patient support during long-term therapy.
Despite limited prior experience with digital health tools, the majority of respondents expressed interest in using a digital support platform.
These findings suggest that well-designed digital health interventions may represent a promising approach to improving symptom management and treatment adherence.
However, the design of such solutions must prioritize usability, accessibility and evidence-based content to ensure patient engagement and trust.
8. Limitations
Several limitations should be considered when interpreting the results of this survey.
First, the sample size remains relatively limited and may not fully represent the diversity of breast cancer patients.
Second, the data collected were self-reported and may be subject to recall bias.
Finally, the cross-sectional design does not allow assessment of longitudinal outcomes or causal relationships.
Further studies involving larger cohorts and prospective follow-up will be necessary to evaluate the clinical impact of digital health interventions in breast cancer survivorship and will be assessed by RESIL.IO in the next coming months.
9. Conclusion
Hormonal therapy remains a cornerstone of breast cancer treatment but is frequently associated with side effects that can negatively impact quality of life and treatment adherence.
The findings of this survey highlight substantial unmet needs in symptom management, information access and lifestyle support among breast cancer survivors.
Digital health platforms such as RESIL.IO may offer scalable, patient-centered solutions to support women throughout long-term hormone therapy treatment.
Future research should focus on evaluating the clinical impact of such digital interventions and their integration into standard oncology care pathways.
References
[1] Roberta Rossa et al. Adherence to adjuvant hormone therapy in breast cancer patients. J Clin Oncol. 2023 Jan 21;30(2):1461-1472.
[2] Partridge AH et al. Nonadherence to adjuvant endocrine therapy in women with breast cancer. J Clin Oncol.2003;21:602–606.
[3]Bassel Odeh et al. Optimizing cancer care through mobile health Support Care Cancer 2015 Jul;23(7):2183-8le health Support Care Cancer 2015 Jul;23(7):2183-8
© 2026 RESIL.IO. All rights reserved.
This document is the exclusive property of RESIL.IO SAS.
Any reproduction, distribution, modification, or use, in whole or in part,
without prior written authorization is strictly prohibited.

